Beginning with hydrogen, and continuing across the periods of the periodic table, we add one proton at a time to the nucleus and one electron to the proper subshell until we have described the electron configurations of all the elements. 
To determine the electron configuration for any particular atom, we can “build” the structures in the order of atomic numbers. Finally, draw diagonal lines from top to bottom as shown. Be sure to only include orbitals allowed by the quantum numbers (no 1 p or 2 d, and so forth). Simply make a column for all the s orbitals with each n shell on a separate row. This chart is straightforward to construct. The arrow leads through each subshell in the appropriate filling order for electron configurations. 
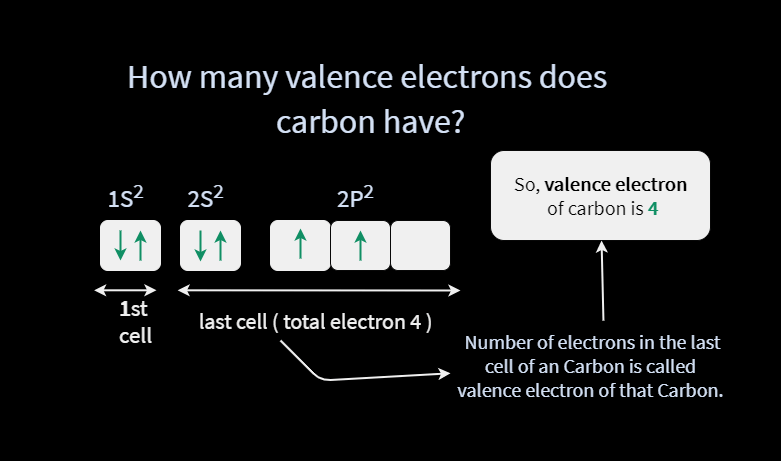
The diagram of an electron configuration specifies the subshell (n and l value, with letter symbol) and superscript number of electrons. The notation 3 d 8 (read “three–d–eight”) indicates eight electrons in the d subshell (i.e., l = 2) of the principal shell for which n = 3.įigure 4.1.2.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
